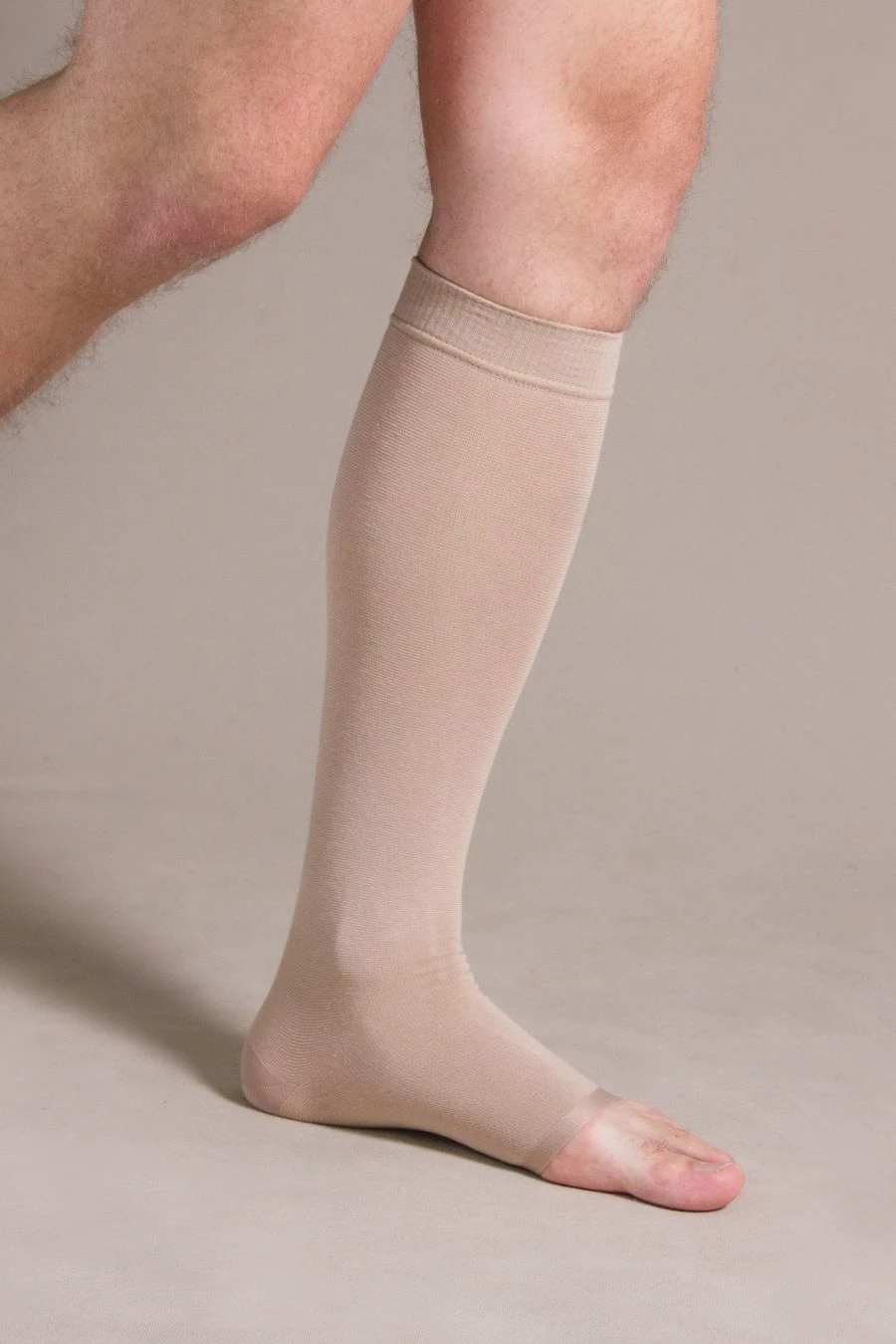

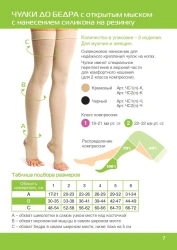
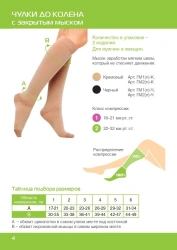
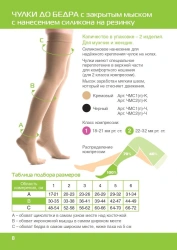
Knee-length compression stockings with open toe are certified medical devices. Registration certificate for medical device FSR 2009/06182 from 29.08.2022 . The products are intended for prevention and treatment of varicose veins of the lower limbs, lymphostasis, post-traumatic complications of the lower limbs, with osteoarthritis and arthritis, with swelling after removal of plaster bandages, as well as for the prevention of thromboembolism in the pre- and postoperative period.Contraindications: -open wounds in the area of the lesion vessel, local skin diseases (dermatitis), allergic reactions to the materials of the stockingSuitable for both men and women, the duration of use of the product is determined by the attending physician.Composition: polyamide 60%, cotton 30%, elastane 10%
















Tell us what you need and get quotes from verified suppliers