

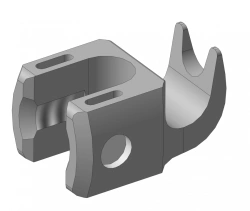
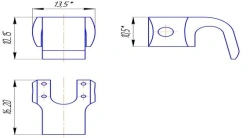
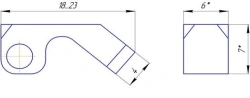
Non-sterile product intended for use in spinal segment fusion to treat anatomical spinal abnormalities typically caused by degenerative disc disease (i.e. DDD) to relieve pressure on pinched nerves and prevent spinal disc slippage. It is a small sized hollow cylinder with holes, made of metal [usually titanium (Ti)], which is implanted between bones or bone grafts of the spine to provide mechanical stability and sufficient space for therapeutic spondylodesis.
Ø4 mm through-holes on the lateral surfaces. Unidirectional harpoon-type spikes on the upper and lower surfaces of the cage. Laser marking of anteroposterior size and height on the product.
Must be sterilised before use.

















Tell us what you need and get quotes from verified suppliers