










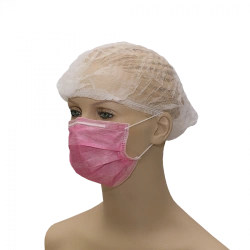
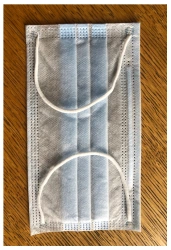
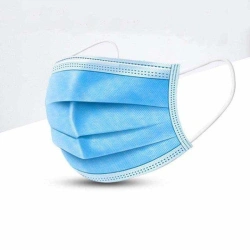
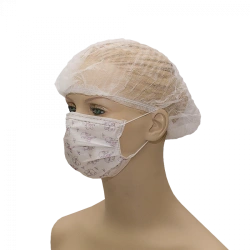
Medical face mask "SORBOTEK" is designed to protect the respiratory tract from dangerous biological objects, the mask is intended for single use.
The mask is used as a barrier means for the population, including when there is a threat of spread of airborne infections. The mask is intended for single use, therefore it is not subject to maintenance and repair.
Medical face mask "SORBOTEK" is non-sterile. Manufacturing material: spunbond, CMC (spunbond/meltblown/spunbond).
The manufacturer guarantees the conformity of the product quality to the requirements of technical specifications if the consumer complies with the conditions and rules of transport, storage and operation.
The warranty period of operation is not set due to the single use of the product. The warranty period of storage of products in the packaging of the manufacturer is 5 years from the date of manufacture.
















Tell us what you need and get quotes from verified suppliers